Somashekar Shetty
Department of Biochemistry, Melaka Manipal Medical College,
Saraswati Udupa
Department of Biochemistry, KMC International Centre and
Laxminarayana Udupa
Department of Pharmacology, Kasturba Medical College, Manipal 576104, India
Introduction
Wound healing is the process of repair that follows injury to the skin and other soft tissues. It is fundamentally a connective tissue response. Initial stages of wound healing involve an acute inflammatory phase followed by synthesis of collagen and other extracellular matrix which are later remodeled to form scar.
Ocimum sanctum (Sanskrit:Tulasi; family: Labiaceae), is found throughout the semitropical and tropical parts of India. Different parts of the plant are traditionally used in Ayurveda and Siddha systems for the treatment of diverse ailments like infections, skin diseases, hepatic disorders and as an antidote for snake bite and scorpion sting (1). A methanol extract and an aqueous suspension of O. sanctum leaves have anti-inflammatory, analgesic and immunostimulatory properties (2). Flavonoids isolated from O. sanctum scavenged free radicals in vitro and showed antilipoperoxidant activity in vivo at very low concentration (3). The free radical scavenging activity of plant flavonoids help in the healing of wounds (4). Low levels of antioxidants accompanied by raised levels of markers of free radical damage play a significant role in wound healing in rats (5).
Free radical scavenging activity is a major mechanism by which O. sanctum products protect against cellular damage (6). Based on studies in rats, O. sanctum may act at various levels in the immune mechanism, such as antibody production, release of mediators of hypersensitivity reactions and tissue responses to these mediators in the target organs in modulating the humoral immune responses (7). Significant inhibition of growth of pathogenic microorganisms was observed in vitro by traditional drugs like O. sanctum, Azardicta indica and Annona squamosa. Healing of the wound by indigenous ointment formulation was comparable with that of nitrofurazone and propamidine cream in mice infected by the organisms (8).
Keeping in view the tremendous pharmacological activities and a wealth of available literature, O. sanctum may be utilized to alleviate the symptoms of a variety of diseases as evident from pre-clinical data (9). Thus it appears that different mechanisms like free radical scavenging, metal chelation as well as immune modulation may act at different levels individually or in combination to bring about the wound healing effects of this medicinal plant.
From another viewpoint using animals for healing it is now universally acknowledged that maggot therapy can be used successfully to treat chronic, long-standing, infected wounds, which have previously failed to respond to conventional treatment. Such wounds are typically characterized by the presence of necrotic tissue, underlying infection and poor healing (10). Maggot therapy employs the use of freshly emerged, sterile larvae of the common green-bottle fly, Phaenicia (Lucilia) sericata and is a form of artificially induced myiasis in a controlled clinical situation. Maggot therapy has the following three core beneficial effects on a wound: debridement, disinfection and enhanced healing (11).

Methods
Plant Material
Leaves of O. sanctum were collected during September from the local areas of Udupi district, Karnataka, India, and were authenticated by Professor Gopalkrishna Bhat, Department of Botany, Poorna Prajna College, Udupi.
A voucher specimen (No. pp531) has been deposited at the Department of Pharmacognosy, College of Pharmaceutical Sciences, Manipal, India.
Preparation of Extracts
Aqueous Extract
Leaves of O. sanctum were dried in shade and powdered. The leaf powder (100 g) was refluxed with 750 ml of double distilled water for 1 h at 75–80_C. It was then cooled and filtered. This was repeated in three trials. The extracts were pooled and evaporated using lyophilizer (12). The yield was about 20–25%.
Alcoholic Extract
75 g of leaf powder was extracted with 700 ml of 95% ethanol in a soxhlet apparatus at 60–75_C. Extract was concentrated by evaporation (12). The yield was about 10–15%. The semisolid extract was dissolved in saline by using gum acacia as a vehicle during the study.
Rats
Healthy Wistar albino rats of either sex and of approximately the same age, weighing between 150–250 g were used for the study. They were individually housed, maintained in clean polypropylene cages and fed with commercially pelleted rat chow (M/s Hindustan Lever Ltd., Mumbai) and water ad libitum. The experimental protocol was subjected to scrutiny of Institutional Animal Ethical Committee for experimental clearance (No. IAEC/KMC/UA/2000).
The animals were divided into five groups of six animals each and were daily administered the extract of O. sanctum via intragastric tube. The extracts were dissolved by using the vehicle gum acacia in normal saline during the study. Two grams of gum acacia was dissolved in 100 ml of normal saline. From this, 10 ml of solution, which contains 200 mg of gum acacia, was used for dissolving 1 g of aqueous and alcoholic extracts. So each ml of solution contains 100 mg of extracts.
Group I: Control group with wound and treated with gum acacia in normal saline
Group II: Test group with wound and treated with aqueous extract (400 mg/kg body weight)
Group III: Test group with wound and treated with aqueous extract (800 mg/kg body weight)
Group IV: Test group with wound and treated with alcoholic extract (400 mg/kg body weight)
Group V: Test group with wound and treated with alcoholic extract (800 mg/kg body weight)
Experimental Procedure
The wounding procedure was carried out using ketamine (1 ml/kg body weight) anesthetized rats in three different wound models. The extract was given daily to the rats orally in the case of incision and dead space wound for 10 days. In the case of excision wound model, the extract was given everyday, till the day of epithelization.
Wound Models
Incision Wound
Two paravertebral incisions (6 cm long) were made through the full thickness of the skin on either side of the vertebral column (13). Wounds were closed with interrupted sutures, 1 cm apart. The sutures were removed on the 7th day. Wound breaking strength (WBS) was measured on the 10th post-wounding day (14).
Determination of Wound Breaking Strength
Rats were secured to the operation table and a line was drawn on either side of the wound 3mm away from the wound. Two allice forceps were firmly applied on to the line facing each other. One of the forceps was fixed, while the other was connected to a freely suspended lightweight polypropylene graduated container through a string run over to a pulley. Water was allowed to flow from the reservoir slowly and steadily into the container. A gradual increase in weight was transmitted to the wound site pulling apart the wound edges. As and when the wound just opened up, the water flow was arrested and the volume of water collected in the container (approximately equal to its weight) was noted. Three readings were recorded for a given incision wound and the procedure was repeated on the wound on the contralateral side. The average reading of the group was taken as an individual value of breaking strength. Mean value gives the breaking strength for a given group.
Excision Wound
A circular piece of full thickness (_500mm2) was cut off from a predetermined area on the back of the rat. Wounds were traced on 1mm2 graph paper on the day of wounding and subsequently on the alternate days, until healing was complete. Changes in wound area were calculated, giving an indication of the rate of wound contraction. Number of days required for falling of eschar without any residual raw wound gave the period of epithelization (15).
Dead Space Wound
These wounds were created by implanting two polypropylene tubes (0.5_2.5 cm2 each), one on either side in the lumbar region on the dorsal surface of each rat. On the 10th post-wounding day, the granulation tissue formed on the implanted tubes was carefully dissected out. The granulation tissues were collected, and dried at 60_C for 24 h. The dry tissue was used for the estimation of hydroxyproline (16), hexosamines (17) and hexuronic acid (18).
Granulation tissue from the other tube was collected in phosphate-buffered saline for the estimation of antioxidant enzymes like superoxide dismutase (SOD) (19), catalase (20), reduced glutathione (GSH) (21) and tissue lipid peroxidation (22).
Acute Toxicity Studies
Healthy rats (n¼6) were orally fed with increasing doses (400 mg/kg to 6 g/kg body weight) of extracts for 14 days. The doses up to 4 g/kg body weight did not produce any toxicity and mortality. Of the maximum-tolerated doses (400 and 800 mg/kg), 10 and 20% were selected for the study.
Statistical Analysis
The results were analyzed using one way analysis of variance (ANOVA) with post hoc Scheffe’s test. P-values 50.05 were considered statistically significant.
Results
Physical Parameters
In the incision wound model, WBS was measured. WBS was significantly increased in the O. sanctum-treated group compared with control with both the extracts and with both the doses. There was not much significant difference in WBS in the dose and type of extracts (Fig. 1A). In the excision wound model, the percent of wound contraction was measured on alternate days. The percentage wound contraction was nearly 100% for the both the doses of alcoholic extract in 16 days while for the control it was 22 days (P50.05). The percentage wound contraction was nearly 100% for the both the doses of aqueous extract in 14 days in comparison with controls (P50.05) (Fig. 2). In the above physiological and pharmacological parameters of wound healing, aqueous extract of O. sanctum was more effective than the alcoholic extract.
Biochemical Parameters
In the dead space wound model, the granulation tissue hydroxyproline level was significantly increased in alcoholic extract 400 mg/kg (P50.01), and in all other types of extract-treated groups compared with control (P50.001). There was no significant difference among different doses of alcoholic extract. When compared with alcoholic extract 400 mg/kg, there was significant increase in hydroxyproline level in aqueous extract 800 mg/kg (P50.01) (Fig. 1B). Hexuronic acid level was significantly increased in all extract-treated groups compared with control (P50.01) and it was highly significant for aqueous 800 mg/kg treated group (P50.001) (Fig. 1C). Hexosamine level was significantly increased in aqueous and alcoholic (400 and 800 mg/kg) extracttreated groups compared with control (P50.05 and -P50.01) (Fig. 1D).
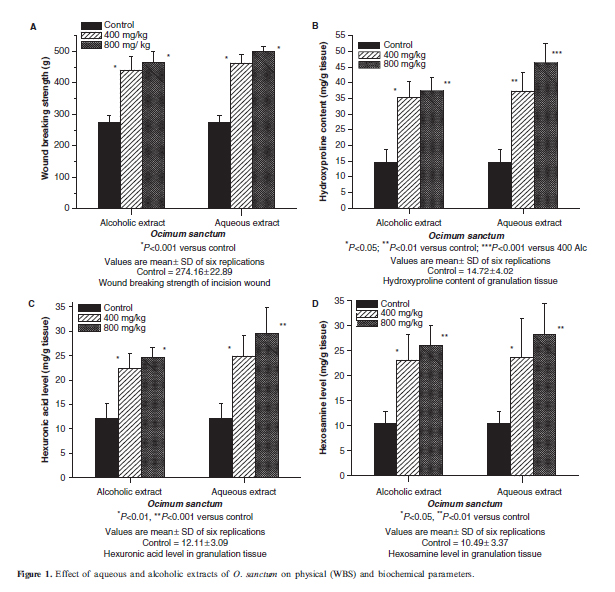
Antioxidant Parameters
SOD activity in granulation tissue was significantly increased in the case of rats treated with alcoholic and aqueous extract 400 mg/kg (P50.05), alcoholic 800 mg/kg (P50.05) and aqueous extract 800 mg/kg (P50.01) when compared with control (Fig. 3A). Catalase level in granulation tissue was significantly increased in the case of alcoholic and aqueous extract 400 mg/kg (P50.05), alcoholic 800 mg/kg (P50.05) and aqueous extract 800 mg/kg treated groups (P50.01), when compared with control (Fig. 3B). Reduced GSH concentration in granulation tissue was significantly increased in rats treated with different extracts (P50.001), when compared with control. There was significant increase in GSH level in aqueous 800 mg/kg treated group when compared with alcoholic 400 mg/kg group (P50.05) in intercomparison statistical analysis (Fig. 3C). Malondialdehyde (MDA) level (which indicates lipid peroxidation) in granulation tissue was significantly decreased in the case of all the extract-treatment groups (P50.001) compared with controls (Fig. 3D).

Discussion
Granulation, collagen maturation and scar formation are some of the many phases of wound healing, which run concurrently, but independent of each other. The use of a single model is inadequate and no reference standard exists that can collectively represent the various phases of wound healing. Hence, three different models have been used in our study to assess the effect of O. sanctum on the various phases of wound healing.
In incision wound, the increase in tensile strength of treated wounds may be due to the increase in collagen concentration and stabilization of the fibers (23). Increase in WBS and role of antioxidants were experimentally proved by Michel and Fredrickson (24). In excision wound, both the extracts showed faster healing compared with control groups and wound contraction rate is faster with aqueous extract as compared with alcoholic extract. The faster wound contraction rate of the aqueous extract may be due to stimulation of interleukin-8, an inflammatory a-chemokine which affects the function and recruitment of various inflammatory cells, fibroblasts and keratinocytes. It may increase the gap junctional intracellular communication in cultured fibroblasts and induces a more rapid maturation of granulation tissue (25). The aqueous extract of O. sanctum increased cellular proliferation and collagen synthesis at the wound site as evidenced by increase in total protein and total collagen contents reflected by hydroxyproline content of granulation tissues.
The glycosaminoglycans are a major component of the extracellular matrix of skin, joints, eyes and many other tissues and organs. In spite of its simple structure, it demonstrates remarkable viscoelastic and hygroscopic properties which are relevant for dermal tissue function. Biological activities in skin are due to its interaction with various binding proteins. Due to an influence on signaling pathways, hyaluronic acid is involved in the wound healing process and scarless fetal healing. In clinical trials, topical application of hyaluronic acid has improved the healing of wound (26). In addition, the mucopolysaccharide hyaluronic acid protects granulation tissue from oxygen free radical damage and thereby stimulates wound healing (27).
Among the glycosaminoglycans, dermatan sulfate and dermatan have also been implicated in wound repair and fibrosis. Their ability to bind and alter protein–protein interactions has identified them as important determinants of cellular responsiveness in development, homeostasis and disease (28). In our study, hexuronic acid and hexosamine concentrations which are the component of glycosaminoglycans were significantly increased with both the extracts and with both the doses when compared with control. The glycosaminoglycans are known to stabilize the collagen fibers by enhancing electrostatic and ionic interactions with it and possibly control their ultimate alignment and characteristic size. Since O. sanctum extracts have increased the levels of these compounds considerably, it is likely that the observed increase in tensile strength was not only due to increased collagen synthesis but also due to its proper deposition and alignment.
Molecular oxygen plays a central role in the pathogenesis and therapy of chronic wounds. Overproduction of reactive oxygen species (ROS) results in oxidative stress thereby causing cytotoxicity and delayed wound healing. Therefore, elimination of ROS could be an important strategy in healing of chronic wounds (29). Therefore, estimation of antioxidants like SOD, catalase and glutathione in granulation tissues is also relevant because these antioxidants hasten the process of wound healing by destroying the free radicals (30). The significant alteration in the antioxidant profile accompanied by the elevated levels of MDA, a marker of free radical damage, may be attributed to impaired wound healing in immunocompromised rats (31). While numerous attempts have been made to identify prognostic biomarkers of wound healing in skin, these have met with limited success. The bioactivity of flavonoids tightly correlated with their chemical structure and action mechanisms, mostly inhibitory on enzymatic systems involved in cellular activations (32). Study of 12 different plant flavonoids, mainly orientin and vicenin of O. sanctum has revealed that their in vitro antioxidant activity works also under in vivo conditions (33).
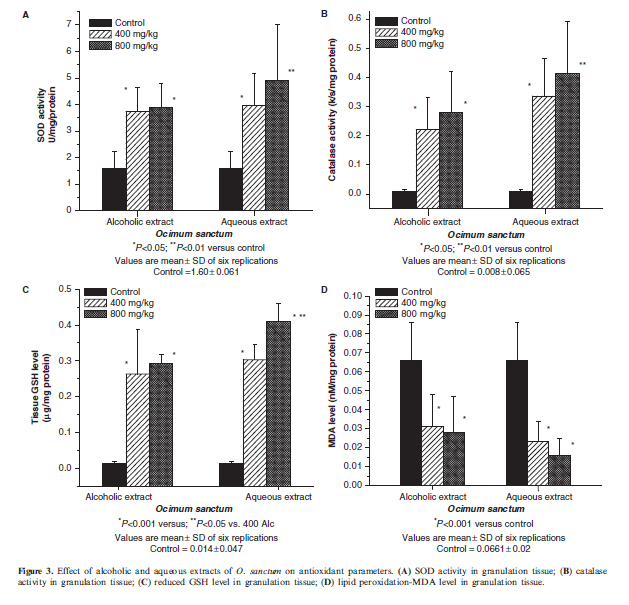
In case of the antioxidant parameters, rats treated with the aqueous extracts of O. sanctum in the dose of 800 mg/kg body weight showed significant increase in the activity of SOD catalase and GSH with a decrease in MDA level in granulation tissue compared with controls. These enzymes are known to quench the superoxide radical and thus prevent the damage of cells caused by free radicals (34). Better collagenation, seen under the influence of this plant extract, may be because of the presence of flavonoids, which is responsible for the free radical scavenging activity which is believed to be one of the most important components of wound healing (35).
In our study on the effect of alcoholic and aqueous extracts of O. sanctum on wound healing (400 and 800 mg/kg body weight), we found that aqueous extract possesses a better effect than alcoholic extract at a dose of 800 mg/kg body weight. Since O. sanctum is ubiquitous and abundantly grown, it could be a fairly economical therapeutic agent for wound management as a prohealer, as well as to control abnormal healing.
References
- Godhwani S, Godhwani JL, Vyas DS. Ocimum sanctum – a preliminary study evaluating its immunoregulatory profile in albinom rats. J Ethnopharmacol 1988;24:193–8.
- Godhwani S, Godhwani JL, Vyas DS. Ocimum sanctum: an experimental study evaluating its anti-inflammatory, analgesic and antipyretic activity in animals. J Ethnopharmacol 1987;21:153–63.
- Uma Devi P, Ganasoundari A, Vrinda B, Srinivasan KK, Unnikrishnan MK. Radiation protection by the Ocimum flavonoids orientin and vicenin: mechanism of action. Radiat Res 2000; 154:455–60.
- Havsteen BH. The biochemistry and medical significance of the flavonoids. Pharmacol Ther 2002;96:67–72.
- Rasik AM, Shukla A. Antioxidant status in delayed healing type of wounds. Int J Exp Path 2001;81:257–63.
- Ganasoundari A, Uma devi P, Rao BS. Enhancement of bone marrow radiation protection and reduction in WR-2721 toxicity by Ocimum sanctum. Mutat Res 1998;373:271–5.
- Mediratta PK, Sharma KK, Singh S. Evaluation of immunomodulatory potential of Ocimum sanctum seed oil and its possible mechanism of action. J Ethnopharmacol 2002;80:15–20.
- Thaker AM, Anjaria JV. Antimicrobial and infected wound healing response of some traditional drugs. Indian J Pharmacol 1986;18:171–4.
- Nigam Y, Bexfield A, Thomas A, Ratcliffe NA. Maggot therapy: the science and implication for CAM part I—history and bacterial resistance. Evid Based Complement Alternat Med 2006;3:223–7.
- Nigam Y, Bexfield A, Thomas A, Ratcliffe NA. Maggot therapy: the science and implication for CAM part II—Maggots Combat Infection. Evid Based Complement Alternat Med 2006;3:303–8.
- Gupta SK, Prakash Jai, Srivatsava Sushma. Validation of traditional claim of Tulsi, Ocimum sanctum Linn. As a medicinal plant. Indian J Exp Biol 2002;40:765–73.
- Suffness M, Douros J. New natural products of interest under development at the National Cancer Institute. Cancer Chemother Pharmacol 1978;1:91–100.
- Ehrlich HP, Hunt HK. Effect of anabolic steroid on tensile strength of a healing wound. Ann Surg 1969;170:203–8.
- Lee KH. Studies on the mechanism of action of salicylates 3. Effect of vitamin A on wound healing retardation of aspirin. J Pharmacol Sci 1968;57:1238–40.
- Morton JJ, Malone MH. Evaluation of vulnerary activity by open wound procedure in rats. Arch Int Pharmacodyn Ther 1972; 196:117–20.
- Neuman RE, Logan MA. The determination of collagen and elastin in tissues. J Biochem 1972;186:549–56.
- Boas NF. Method for the determination of hexosamines in tissues. J Biol Chem 1953;204:553–5.
- Bitter T, Muir H. A modified uronic acid carbazole reaction. Anal Biochem 1962;4:330–2.
- Kakkar Poonam, Das Ballabh, Vishwanathan PN. A modified spectrophotometric assay of superoxide dismutase. Indian J Biochem Biophysics 1984;21:130–2.
- Aebi HE. Methods of Enzymatic Analysis, 3rd ed, Vol. III, New York Academic Press; 1974;273–84.
- Beutler E, Duron O, Kelly BM. The improved method for the determination of blood glutathione. J Lab Clin Med 1963;61: 882–8.
- Konings AWT, Drijiver EB. Measurement of lipid peroxidation. Radiat Res 1979;80:494–501.
- Udupa AL, Kulkarni DR, Udupa SL. Effect of Tridax procumbans extracts on wound healing. Int J of Pharmacognosy 995;33:37–40.
- Michel JW, Fredrickson. Wound healing—oxygen free radicals and wound healing. Clin Plast Surg 1990;17:1473–83.
- Moyer KE, Saggers GC, Ehrlich HP. Effects of interleukin-8 on granulation tissue maturation. J Cell Physiol 2002;193:173–9.
- Weindl G, Schaller M, Korting HC. Hyalronic acid in the treatment and prevention of skin diseases: molecularbiological, pharamaceutical and clinical aspects. Skin Pharmacol Physiol 2004;17:207–13.
- Bayliss MT. Proteoglycans: structure and molecular organisation in cartilage. In: Hukins DWL (ed). Connective Tissue Matrix. London: Macmillan, 1984, 55.
- Trowbridge JM, Gallo RL. Dermatan sulphate—new functions from an old glycosaminoglycan. Glycobiol 2002;12:117R–25R.
- Dissemond J, Goos M, Wagner SN. The role of oxidative stress in the pathogenesis and therapy of chronic wounds. Hautarzt 2002;53:718–23.
- Halliwell B, Gutteridge JM, Grootveld M. Methods for the measurements of hydroxyl radicals in biomedical systems; deoxyribose degradation and aromatic hydroxylation. Methods of Biochem Anal 1988;33:59–90.
- Gupta A, Singh RL, Raghubir R. Antioxidant status during cutaneous wound healing in immunocompromised rats. Mol Cell Biochem 2002;241:1–7.
- Ielpo MT, Basile A, Miranda R, Nappo C. Immunopharmacological properties of flavonoids. Fitoterapia 2000;71(Suppl 1):101s–9s.
- Shimoi K, Masuda S, Shen B, Furugori M, Kinae N. Radioprotective effects of antioxidative plant flavonoids in mice. Mutat Res 1996;350:153–61.
- Liu F, Ooi VEC, Chang ST. Free radical scavenging activities of mushroom polysaccharide extracts. Life Sci 1997;60(10):763–8.
- Devipriya S, Shyamaladevi CS. Protective effect of quercetin induced cell injury in the kidney. Indian J Pharmacol 1999;31:422–4.



